I am Dr. Minotti, a regenerative medicine specialist. I have spent the past 20 years exploring new and better ways to treat the human body. I help frustrated patients find freedom from pain and restore their quality of life so they can do the things they love.
Research exploring the physiology of how the body actually heals has allowed innovative advancements in treatment of musculoskeletal conditions. Regenerative Orthopedics helps bodies mend themselves, providing real results for people who have been living — until now — with few treatment options.
Tendon injury
The complex structure of ligaments and tendons that support our joints provides stability. They are composed of collagen, the most abundant protein in the body. Their tough, collagenous structure gives the joint strength. This structure, however, also causes these tissues to have poor blood supply, making the joint prone to chronic conditions.
Tendons are bands of tissue that connect muscles to the bones. Overuse is the most common cause of injury, resulting from repetitive motion that causes the tendons to become damaged. For example, the tendon attaching the muscle to the epicondyle, or protrusions on the elbow, often become inflamed, creating a condition called “Tennis Elbow” or “Golfer’s Elbow.” Symptoms of elbow injuries include pain, stiffness, tingling or numbness, and pain when gripping or lifting.
Tennis Elbow and Golfer's Elbow are common examples of repetitive motion activities that often result in inflammation of the tendons in the elbow. Overhead pitching in young athletes is also a common cause of tendon strain. Poor technique and lack of warm-up can contribute to this condition. Muscles and tendons are like a piece of taffy: when warm, they stretch and are pliable; when cold, they are rigid and prone to injury. The resulting inflammation, although painful, is your body's method of rushing healing cells to the site of injury.
Damaged tendons transmit pain impulses to the brain. Through a subconscious reflex, the surrounding muscles go into a painful spasm in an attempt to stabilize the joint. Often, painful knots appear in the surrounding muscles. Muscles become tight and painful as they try to compensate for the weak and damaged underlying structures. Spasm in the muscles further decreases the blood flow and nutrient supply to the tissue, contributing to increase in the breakdown. This chronic tension leads to deterioration of the tendon attachments and tendinosis, often resulting in chronic tendon degeneration.
Why tendons do not heal well
Different tissues have different blood flow and different metabolic rates. Where blood supply is low, degeneration often occurs due to low oxygen levels and inadequate supply of nutrients and growth factors needed for healing. Inadequate blood flow causes injuries to heal slowly and generally incompletely. Areas with poor circulation are also difficult for stem cells to reach and for cell signaling to be effective, key elements in the Emergency Response Team our body uses to heal.
Tendons, like other collagenous tissue in the joint, have extremely poor circulation. A single molecule of collagen, the primary protein that constitutes tendons, takes about 100 days to fully mature. Tendons also have a slow rate of metabolism, or cellular turnover rate. When injured, tendons take a long time to adapt to stimulation in their environment that signal an injury to the tissue. Consequently, the body's Emergency Response Team is slow to respond. Tissue building nutrients are slow to transport to the site of injury. A muscle, on the contrary, can hypertrophy or atrophy within a few days because it has a higher rate of metabolism.
Tendon structure
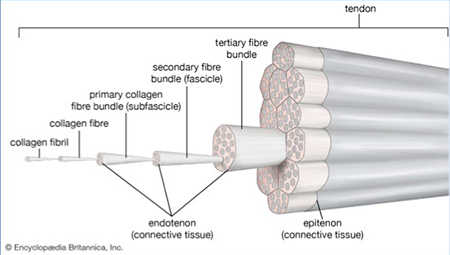
Tendons are bands of dense connective tissue composed of collagen that connect muscles to the bones, transmitting the mechanical force of muscle contraction to move the joint. They have one of the highest tensile strengths found in the body’s soft tissue. A tendon is composed of dense fibrous connective tissue made up primarily of collagenous fibers. Primary collagen fibers consist of bunches of collagen fibrils that form the basic units of a tendon. Groups of primary fibers form secondary fiber bundles that, in turn, group together to form tertiary fiber bundles, together creating the tendon unit (see image).
Spindle-shaped tenocytes (fibrocytes) and tenoblasts (fibroblasts) are the primary cell types in tendons. Mature tendon cells (tenocytes) are found throughout the tendon structure, usually anchored to collagen fibers. Spindle-shaped immature tendon cells (tenoblasts) mature into tenocytes. Tenoblasts are typically clustered and free from collagen fibers. They are highly proliferative in nature and important units in the synthesis of collagen and other extracellular matrix components.
Tendinopathy
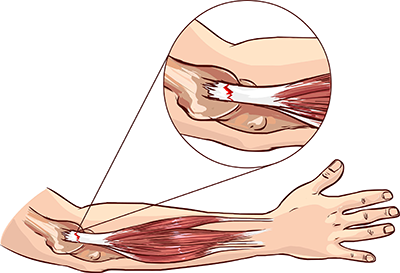 Both tendinopathy and tendonitis are caused by overuse or sudden stress on a tendon. Aging and lack of muscle tone can also play a role. “Tendinopathy” is derived from the Greek term “pathos” which indicates a disease or disorder. Although often used interchangeably, tendinosis is the more precise term for a condition showing degeneration of tissue with “tendinopathy” encompassing a broader range of disorders.
Both tendinopathy and tendonitis are caused by overuse or sudden stress on a tendon. Aging and lack of muscle tone can also play a role. “Tendinopathy” is derived from the Greek term “pathos” which indicates a disease or disorder. Although often used interchangeably, tendinosis is the more precise term for a condition showing degeneration of tissue with “tendinopathy” encompassing a broader range of disorders.
Tendinopathy, or more precisely tendinosis, is a degeneration of the collagen protein that forms the tendon. Tendons may develop small tears or the collagen fibers. Normally aligned in straight segments that glide easily over each other, fibers can become disorganized. This physiological dysfunction causes further damage to the tissue and can, in severe cases, result in a rupture. Scar tissue generally develops causing further biomechanical restrictions. Biomechanical dysfunction is common in underlying conditions such as tennis elbow and swimmer’s shoulder. Rupture to the Achilles heel is not uncommon due to the stress placed on this tendon. Partial ruptures that have a portion of the tendon intact can often be treated non-surgically.
Further distinction between acute and chronic rupture can also be made: an acute tendon rupture is a one-time event that can result in immediate pain and decreased function of the affected joint and may be followed by swelling or bruising. An acute rupture is typically recognized and typically treated within 2 weeks of injury. Chronic tendon ruptures may result from:
- A partial rupture that slowly worsens over a prolonged period, or
- An acute rupture that goes untreated for several weeks (many experts believe this time varies between 4 to 6 weeks, depending on the tendon).
Flint JH, Wade AM, Giuliani J, Rue JP. Defining the terms acute and chronic in orthopedic sports injuries: a systematic review. Am J Sports Med. 2014 Jan;42(1):235-241. doi: 10.1177/0363546513490656. Epub 2013 Jun 7.
Doctors previously thought that tendinosis was an eventual result of tendonitis. But after research examination of samples of injured tendons under a microscope, most now believe it’s the other way around — tendonitis is an eventual result of tendinosis. This relatively new understanding about the underlying causes and progression of tendinosis has resulted in changes to treatment approaches.
Bass E. Tendinopathy: why the difference between tendinitis and tendinosis matters. Int J Ther Massage Bodywork. 2012;5(1):14-17.
Abate M, Silbernagel KG, Siljeholm C, et al. Pathogenesis of tendinopathies: inflammation or degeneration?. Arthritis Res Ther. 2009;11(3):235.
Treatment options
Common treatment for tendon injury has been over-the-counter nonsteroidal anti-inflammatory drugs (NSAIDs), such as ibuprofen. Remember, however, that doctors no longer think that tendon inflammation plays a role in the development of tendinosis. The inverse is now believed to be the actual progression: small tears found in tendinosis lead to the inflammation in tendinitis. With better understanding of the relationship between inflammation and tendinosis, many doctors now question this treatment approach.
Growing evidence shows that NSAIDs may actually slow down the recovery process. A study from 2013 found that diclofenac and corticosteroid injections actually slow down the rate of new tendon cell growth. An earlier study from 2004 found that ibuprofen has a similar effect on Achilles tendon cells in mice.
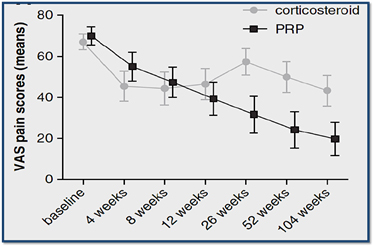
Cortisone has traditionally been another popular treatment for tendon conditions. Multiple studies, including research published in The American Journal of Sports Medicine, show that corticosteroids allow for pain relief by reducing inflammation. The results, however, are short term. Studies also show that cortisone can cause further damage to tissue, resulting in long term degeneration. A study published in 2010 showed the detrimental long-term effect of corticosteroids and their inferior effectiveness in pain relief when compared to the Regenerative Treatments, such as Platelet Rich Plasma (PRP) Injections.
The corticosteroid group performed better initially and then declined, whereas the PRP group progressively improved. This result is due to the fact that PRP treats the root cause of the condition rather than masking the symptoms.
The negative impact of steroids and NSAIDs is partially due to the fact that tendinopathies are not an inflammatory condition. Corticosteroids are catabolic. In other words, they lead to the breakdown of tissue structure. These substances also inhibit cell growth and potentially interfere with immunity. Learn more
Cortisone temporarily stops the pain of injured joints by reducing inflammation. The body heals by Inflammation. When a person sprains an ankle, the persistent swelling for one to three days is the body’s attempt to heal the area. If you want to stop healing, just stop inflammation. This is exactly what cortisone does... The inflammatory cascade that creates healing, is inhibited by cortisone as it minimizes the migration of the immune cells to the injured area. Healing is sacrificed healing for pain control. Elbow Tendonitis. Peerbooms JC, Am J Sports Med 2010. INSAIDs.
Regenerative Orthopedics and tendon Injury.
Research exploring the physiology of how the body actually heals has produced innovations in treatment of musculoskeletal conditions. Regenerative Orthopedics helps bodies mend themselves, providing results for people who have been living — until now — with few treatment options. Bone Marrow Stem Cell Therapy and PRP injections are non-surgical procedures designed to accelerate the repair of tissue in healing of acute and chronic musculoskeletal injuries. They provide growth factors, nutrients, and a healthy cellular environment to promote healing and tissue repair in body areas where blood flow and stem cells cannot easily access. This concept is particularly relevant for joints, ligaments, tendons and even discs.
Platelet Rich Plasma Injections PRP is one of the first Regenerative Therapies for treatment of musculoskeletal conditions, using your own blood cells in concentrated doses to stimulate the natural healing process. It is also effective in reducing joint inflammation, helping to reverse the degenerative effects of osteoarthritis. Results of a study published in the March 2013 issue of Science Daily demonstrate the efficacy of Platelet Rich Plasma (PRP) on tennis elbow, showing continued improvement over an extended period of time.
Bone Marrow Aspirate Concentrate (BMAC) BMAC is a form of Stem Cell Therapy that uses bone marrow tissue from the hip. Research examing the biological mechanisms of cellular health and tissue regeneration is extensive, particularly the role of platelets, stem cells, and natural growth factors in the blood. BMAC Stem Cell Therapy, involves harvesting healthy cells, including stem cells, and precisely injecting them through the use of ultrasound guidance to the injured area. With the needed growth cells, injured tissues begin to repair and heal.
Injured cells in your skin, bones, muscles, ligaments, tendons, and joints send out signals. Stem cells respond to those signals, migrating to the injured tissue and releasing proteins that nourish and stimulate your own cells' regeneration processes.
Natural forces within us are the true healers of disease. ― Hippocrates
Mesenchymal stem cells
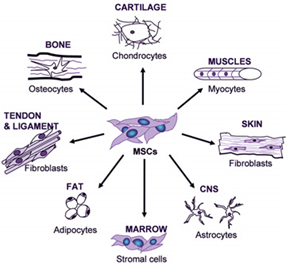
As researchers discover more about the physiological processes involved in tissue repair and regeneration, physicians are able to develop more effective, innovative procedures. Mesenchymal stem cells are leading the way for new and better orthopedic treatment.
Mesenchymal stem cells are multipotent, or have the ability to develop into a wide variety of cells, and are highly proliferative. Chondrocytes, cartilage producing cells, are one of the many cells of differentiation that are beneficial for the treatment of osteoarthritis.
In addition to their function as building blocks for development of new tissue, scientists are also discovering that MSCs mediate the body’s signaling process, or system of cell communication to activate localized stem cells and signal healing cells to the site of the injury.
Mesenchymal stem cells that differentiate into cytokines, small proteins or peptides, mediate the immune system by aiding in cell to cell communication to stimulate the movement of cells toward sites of injury. These cells also reduce inflammation by inhibiting proliferation of lymphocytes.
MSCs play a large role in mediating the signaling process, directing traffic of all the players in our body's Emergency Medical Team that rush to the scene to stimulate our innate regenerative potential, regenerating millions of cells each minute. As our body's natural system becomes overwhelmed, BMAC can enhance our body's natural process to restore tissue, eliminate pain, and revive range of motion.
Signaling
Signaling is a process our body uses to set in motion the body's Emergency Response Team, signaling to cells throughout the body the message to rush healing elements to injured tissue. Cells can detect what is happening in their environment and respond in real time to cues from other cells. They perceive cellular needs and respond with proteins, growth factors, nutrients, and stem cells to meet this emergency, stimulating the repair process. Cell signaling is part of a communication process governing basic cellular activity throughout the body. Mesenchymal stem cells are the captain of this process.
Injected stem cells dock next to your damaged cells and release growth factors, cytokines, and chemokines which help to:
- Stimulate cells to regenerate tissue
- Activate T-cells to secrete proteins
- Open blood vessels and form new ones
- Inhibit your inflammation
- Move cells out of your blood vessels and into the tissues surrounding them
- Regulate your immune system
Our bodies are amazing, constantly replenishing cells to attain maximum wellness and to heal injury and degeneration. Sometimes, however, the rate of replenishment cannot keep up with the demand. The goal of Regenerative Medicine is to amplify this natural repair system to keep pace with these demands.
Leader in Regenerative Orthopedics
Dr. Minotti is Board Certified and specialty trained in Neuromusculoskeletal Medicine and Osteopathic Manipulative Medicine by the American Board of Neuromusculoskeletal Medicine. He has extensive additional training in Regenerative Orthopedics. Dr. Minotti also serves as an adjunct professor at University of North Texas Health Science Center.
Dr Minotti began performing Regenerative Procedures in 2007 and has successfully treated thousands of patients. He continues to study with the world’s leading physicians to find better, more effective methods of treatment for his patients. The use of Orthobiologics in treatment of musculoskeletal conditions is an emerging field with evidence of its success accelerating rapidly. Research examining the biological mechanisms of cellular health and tissue regeneration is extensive, particularly the behavior of platelets, stem cells, and natural growth factors in the blood.
Regenerative Orthopedics addresses the root cause of your pain rather than merely treating the symptoms, effectively bridging the gap between invasive surgery and medications.
Research
Osteoarthritis-derived chondrocytes are a potential source of multipotent progenitor cells for cartilage tissue engineering
https://www.ncbi.nlm.nih.gov/pubmed/27644879
Osteoarthritis prevention through meniscal regeneration induced by intra-articular injection of meniscus stem cells
https://www.ncbi.nlm.nih.gov/pubmed/23461527
Mesenchymal stem cells as a potent cell source for articular cartilage regeneration
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC4559256
Basic science of osteoarthritis
https://www.ncbi.nlm.nih.gov/pubmed/27624438
Mesenchymal stem cells as a potent cell source for articular cartilage regeneration
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4131275/
Mesenchymal stem cells in the treatment of traumatic articular cartilage defects: a comprehensive review
http://arthritis-research.biomedcentral.com/articles/10.1186/s13075-014-0432-1